
Trend Notes - Scottish Moths
Published: 2019
Prepared using evidence from the National Moth Recording Scheme (NRMS) and the Rothamsted Insect Survey (RIS) by Tom Brereton1 , Emily Dennis1 , Richard Fox1 , Byron Morgan2 , Tom Prescott1 , Chris Shortall3 and Simon Foster4 Affiliations1 Butterfly Conservation;2 University of Kent3 Rothamsted Research;4 Scottish Natural Heritage

Key points
- Of the 558 macro-moth species on the Scottish list, abundance and/or occupancy trends are presented for 244 species (44% of the total).
- The species for which it was possible to produce trends, were typically common and widespread species, whereas rare and range-restricted species are underrepresented due to a lack of sufficient data for the analysis methods used. • Moth abundance has almost halved (46% decline) over the last 25 years, with more ‘significantly decreasing’ than ‘significantly increasing’ species (58 versus 5). However, a moth occupancy indicator from NMRS distribution data, suggests that moth occupancy has increased over the last 25 years by about 16%, with similar numbers of ‘significantly decreasing’ and ‘significantly increasing’ species (51 versus 60).
- It seems likely that summer warming is an important factor driving northward range expansions and corresponding increases in occupancy, whilst this is being countered for some species by negative impacts from land management practices and habitat changes, together with warmer and wetter winters, leading to population declines.
- Further research is needed to understand the drivers of recent changes in moth populations in Scotland.
- Moths make up an important part of Scotland’s biodiversity with about 1,300 species known and have high potential to be used as an indicator to measure progress in conserving Scotland’s biodiversity. There are 99 moth species identified as Priorities on the Scottish Biodiversity List, with conservation delivery programmes planned or underway for 21 priority species, in conjunction with BCS. Five case studies detail some of the action underway for moths in Scotland.
Introduction
Moths are an important part of Scotland’s biodiversity with around 1,300 species known (the total for the British Isles being in the region of 2,600 species), which represents approximately 3% of species (from an estimated 46,000 in Scotland, Usher 2002). Of the 1300 species, 558 are macro moth species (Leverton, R. 2018; Scottish Macros List 2017). The Rothamsted Insect Survey (RIS) and National Moth Recording Scheme (NMRS) databases hold records for 627 (including 112 micro moths) and 563 Scottish moth species respectively. Moths can play an important role in telling us about the health of our environment, since they are widespread and found in many different habitats, and have high sensitivity to environmental changes. Their value as part of the food chain, and in providing pollination services (Macgregor et al., 2015; Hahn & Brühl, 2016) as well as in providing cultural services1 , makes moths useful indicator species. Monitoring their numbers and distribution can provide clues to changes in our own environment, such as the effects on wildlife of new farming practices, pesticides, air pollution and climate change.
Moths have been studied scientifically for over 300 years and rigorous recording has been well developed in Scotland since the 1960s, particularly by the network of standardised traps formed within the RIS coordinated by Rothamsted Research and by volunteer recorders contributing to the NMRS, established in 2007 and coordinated by Butterfly Conservation.
A network of light traps as part of the RIS was established in 1968, with up to 57 traps generating data for Scotland over the series and 14 active in 2014 and the traps operating throughout the year. The number of traps monitored per annum has remained broadly stable over the series. The data were used to compile a composite measure2 of moth abundance for Scotland for the period 1975 to 2004 (Scottish Natural Heritage, 2011). The RIS is complemented by a large body of independent moth trapping by volunteer recorders, especially since the 1970s, the data from which is now subsumed into the NMRS. Recent developments in analysis methods bring possibilities in using such recording data to derive robust occupancy (distribution) trends (Isaac et al., 2014; Dennis et al., 2017).
Recent work (Fox et al., 2013; Fox et al., 2014) has shown that some moth populations have undergone large population declines and or range changes across Great Britain and correlative studies and extrapolation from related taxa suggest that agricultural intensification, changes in woodland management and climate change are likely to be major drivers (Fox, 2013). We know relatively little of the status of moths in Scotland, though we do know that some populations are declining (Scottish Natural Heritage, 2011; Fox, 2013). The strength of evidence available to assess the drivers of change in moth species is substantially lower than that available for other well monitored animal groups and has been highlighted as a key knowledge gap (Burns et al., 2016) and there is a need for further research to interpret the changes described in this Trend Note. Climate change is likely to be driving the accelerating northward range expansions recorded for moths in GB as a whole (Mason et al., 2015) and in Scotland (Dennis et al., 2019) and therefore the corresponding increases in occupancy, whilst being countered for some species by negative land management and other environmental changes.
1 The non-material benefits people obtain from ecosystems are called ‘cultural services'. They include aesthetic inspiration, cultural identity, sense of home, and spiritual experience related to the natural environment. Typically, opportunities for tourism and for recreation are also considered within the group. Cultural services are deeply interconnected with each other and often connected to provisioning and regulating services: For example, small scale fishing is not only about food and income, but also about fishers’ way of life. In many situations, cultural services are among the most important values people associate with Nature – it is therefore critical to understand them. 2 A composite index is one that combines data from a number of species into one line.
Moth conservation in Scotland is delivered within the framework of the Scottish Biodiversity Strategy outlined in three strategy documents '2020 Challenge for Scotland's Biodiversity', 'Scotland's Biodiversity: It's in Your Hands' and the ‘Scottish Biodiversity Strategy – Route Map to 2020’. The Route Map to 2020 contains a number of priority projects which will help secure the conservation and enhancement of nature and landscapes. Butterfly Conservation Scotland (BCS) is the NGO leading on moth conservation in Scotland, and has identified approaches, priority species and landscapes, as part of its 2025 Conservation Strategy. This trend note describes Scottish moth populations, providing the latest information on long-term changes in their abundance and distribution. It also reviews efforts to conserve moths in Scotland.

Scottish Moths
UK moths only found in Scotland include the New Forest Burnet Zygaena viciae argyllensis, Slender Scotch Burnet Zygaena loti scotica, Netted Mountain Moth Macaria carbonaria, Cousin German Protolampra sobrina, Broad-bordered White Underwing Anarta melanopa, Pine-tree Lappet Dendrolimus pini, Scotch Annulet Gnophos obfuscates, Rannoch Brindled Beauty Lycia lapponaria and the micro moths Mountain Ermel Ethmia pyrausta, Mountain Pearl Udea uliginosalis and Scotch Crest Dichomeris juniperella. In addition, many subspecies are endemic to Scotland, such as the subspecies jocelynae of the Narrowbordered Five-spot Burnet Zygaena lonicerae, more commonly referred to as the Talisker Burnet.
Some moths have their UK distribution centred on Scotland, such as Sword-grass Xylena exsoleta and Slender-striped Rufous Coenocalpe lapidata, whilst others, such as the Welsh Clearwing Synanthedon scoliaeformis, have nationally important populations within Scotland. Many species have a northern edge to their British range or become more coastal within Scotland, such as Cinnabar Tyria jacobaeae.
Substantial range changes are taking place, for example the Chamomile Shark Cucullia chamomillae has recently been found 120km north-east of its previous Scottish record (Leverton, 2005), and there have been many new colonists to Scotland in recent years, such as Blair’s Shoulder-knot Lithophane leautieri (Agassiz, 2004), Beautiful Snout Hypena crassalis, and the micros Thistle Ermine Myelois circumvoluta and Gold Triangle Hypsopygia costalis. A summary of recently discovered moths recorded in Scotland is given in Table 1.
Table 1. Macro-moths recently discovered in Scotland
Data sources and methods
The Rothamsted Insect Survey (RIS) abundance data
The RIS monitors nocturnal moth populations through a UK-wide network of standardised and automated light-traps that are run every night of the year (Storkey et al., 2016). Moths are identified and counted by amateur experts and /professional scientists, before the records are computerised. Compared to some other trap designs, Rothamsted light-traps tend to capture small (Leinonen et al., 1998), but consistent samples (Taylor & French, 1974) which makes them best suited for monitoring common and widespread species, for which catches are large enough for statistical analysis (Conrad et al., 2006). The small samples obtained are practical to handle without harming local moth populations (Williams, 1952).
Across the RIS network in Scotland, 634 species have been recorded from 57 sites. The dataset covers 1965-present, though in this analysis we focus on the period 1975-2014, with 1975 being the start year of a previous moth indicator for Scotland using RIS data and 2014 being the last year of available data at the time of analysis. In addition, trends for the period 1990-2014 and 2004-2014 were calculated, to enable comparison with occupancy trends from NMRS data.
Annual indices of abundance were derived from weekly trapping records from standard RIS light traps (Williams, 1948; Woiwod & Harrington, 1994). Of the 606 species sampled from 45 sites since 1975, 176 moth species (including one micro-moth) were identified as having sufficient data for trend analysis, using the criteria identified by Conrad et al. (2006) along with some additions (e.g. removing species with potential for misidentification and substantial changes from low sample sizes).
Moth trends were assessed using a Poisson generalised linear model (GLM), accounting for site and year effects. Trends are assigned into categories as used in Defra species indicators and official species statistics.
Multi-species indices were constructed using the geometric mean (Buckland et al., 2005). Composite linear trends were estimated from the indicator, and a smoothed indicator was estimated by applying a generalised additive model (GAM) to the composite index. Confidence intervals for the linear trends and smoothed indicator were calculated using a simulation approach.
National Moth Recording Scheme (NMRS) distribution data
Scottish species occurrence records for macro-moths (here defined as Lepidoptera families: Hepialidae, Cossidae, Zygaenidae, Limacodidae, Sesiidae, Lasiocampidae, Saturniidae, Endromidae, Drepanidae, Geometridae, Sphingidae, Notodontidae, Erebidae, Nolidae and Noctuidae) were obtained from the NMRS database. These were collated from volunteer observers during recording and data collation activity organised by the Biological Records Centre from the late 1960s to early 1980s and by Butterfly Conservation from 2007 onwards (Heath & Emmet, 1983; Hill et al., 2010). Analysis focussed on occurrence data to produce species-level and composite trends. For Scotland, 1,207,836 moth records were available for 563 species for 1970-2014 considered from 10,594 Scottish 1-km grid squares and 41 vice-counties. However, it was that sufficient data were available for trend analysis for 230 species from a baseline set at 1990. The criteria chosen by expert judgement being: first recorded before 1995, recorded in at least 15 years of the 25 year period, and having an average of at least 30 records per year.
These data were not collected using a standardised protocol, but instead are a collation of unstructured biological observations collected by a large network of volunteer recorders. Such data tend to contain many forms of sampling bias, making it challenging to detect genuine signs of change (Tingley & Beissinger, 2009; Hassall & Thompson, 2010; Isaac & Pocock, 2015). Two main approaches have been developed and applied to Lepidoptera: Bayesian occupancy modelling (Isaac et al., 2014) and a classical approach (Dennis et al., 2017). Using either occupancy approach, for a given target species records of all other species are used to provide non-detection (absence) records. In doing so, occupancy and detection probabilities were estimated. The classical occupancy approach describes variation in occupancy across sites in terms of a set of covariates, chiefly latitude and longitude in this instance. From the NMRS data, species trend categories and an occupancy indicator were derived using the same methods as used for RIS abundance data.

Changes in Scottish moths
Abundance trends
In each time period assessed, the majority of species showed no overall significant change, whilst the number of species in this category progressively increased as the time series shortened (48% of species showed no significant change1975-2014; 64% 1990-2014; 87% 2005-2014). In each time period, there were more significantly decreasing than significantly increasing species, this being particularly marked over the 25 year period from 1990 (58 species decreasing versus 5 species increasing).
The most rapidly increasing species from 1990-2014 were (>500% increase, most rapid first) were: Pine Carpet Pennithera firmata, Clouded Border Lomaspilis marginata, Dun-bar Cosmia trapezina, Angle Shades Phlogophera meticulosa, Early Tooth-striped Trichopteryx carpinata, Small Phoenix Ecliptopera silaceata, Large Yellow Underwing Noctua pronuba and Yellow-line Quaker Agrochola macilenta. Of the top 20 most rapidly increasing species, 16 of the 20 (80% of the total) are associated with woodland habitats, 15 of these being widespread species, suggesting that common woodland moths may be doing well in Scotland. Of the 10 moths that were increasing most rapidly in abundance in the previous assessment (Scottish Natural Heritage, 2011), trends were calculable for six species. Of these, five show a more positive trend than previously, whilst one has a worsening trend (though the trend for that species still positive).
The most rapidly declining species from 1990-2014 were (>90% decline, most extreme decline first) were: Pale Eggar Trichiura crataegi, Anomalous Stilbia anomola, Grass Rivulet Perizoma albulata, Broom Moth Ceramica pisi, Brown-line Bright-eye Mythimna conigera, Mouse Moth Amphipyra tragopoginis, Common Wainscot Mythimna pallens, Diamond-back Moth Plutella xylostella and Black rustic Aporophyla nigra. Of the top 20 species in most rapid decline, 13 were associated with semi-natural habitats, seven with a broader range of habitats including intensive farmland and plantation woodland and urban areas, whilst one is a common migrant. Eight of the 20 species are associated with moorland, suggesting moths may be declining disproportionately in this habitat. Of the 10 moths that were declining most rapidly in abundance in the previous assessment (Scottish Natural Heritage, 2011), trends were calculable for eight species with sufficient data. Of these, seven have declined to a greater degree than in the previous assessment, whilst one species has an improved trend (though the trend for that species is still negative).
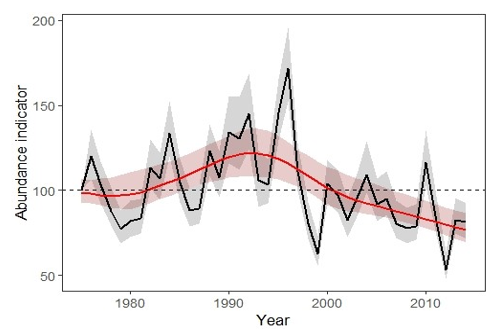
The composite indicator (based on 176 species) of moth abundance dropped by a fifth from 1975-2014 (Figure 1). Within this 40-year time series, abundance increased significantly in the early part of the series (by 22%), subsequently declining from the early 1990s. Trends were assessed for four time periods (1975-2014, 1975-1990, 1990-2014 and 2005-2014). Over the 40-year period, moth abundance declined significantly by 20%. The decline was mostly in the latter part of the series (-46% 1990-2014), with moth abundance increasing significantly earlier in the series (+22% 1975-1990). Moth abundance declined significantly by 16% from 2005-2014.
Using RIS data, the overall significant decline in Scottish moth abundance of 20% for the 40-year period 1975-2014, is similar to the overall decline in abundance of 28% for UK moths, for the 40-year period 1968-2007 described by Fox (2013). These results indicate that Scottish moths may be declining in abundance at a similar rate to those across the UK as a whole, though one should note the time period differences.
The results suggest that any benefits for established species brought about by the warming summer climate, are being cancelled out by warmer and wetter winters and negative land management practices in some habitats, and habitat changes leading to an overall population decline. Though the results of these analyses give clear signals, they require further investigation, given the relatively small number of traps that make up the RIS network in Scotland.
Using RIS data, the overall significant decline in Scottish moth abundance of 20% for the 40- year period 1975-2014, is similar to the overall decline in abundance of 28% for UK moths, for the 40-year period 1968-2007 described by Fox (2013). These results indicate that Scottish moths may be declining in abundance at a similar rate to those across the UK as a whole, though one should note the time period differences.
Occupancy trends
Occupancy measures the distribution of moths across Scotland. Changes in occupancy can reflect species expanding their distributions into new regions or becoming more widespread within their established range. In each time period assessed, the majority of species showed no overall change in occupancy. The proportion of species showing no change in trend was higher in the most recent time period (52% of species 1990-2014; 82% 2005-2014). In each time period, there were similar numbers of increasing and decreasing species.
The most rapidly increasing species from 1990-2014 (>200% increase, most rapid first) were: Spruce Carpet Thera britannica, Grey Dagger Acronicta psi, Lempke's Gold Spot Plusia putnami, Frosted Orange Gortyna flavago, Clouded Silver Lomographa temerata, Satin Beauty Deileptenia ribeata, Chestnut Conistra vaccinii, Merveille Du Jour Griposia aprilina and Grey Arches Polia nebulosa. Of the top 20 species increasing most rapidly, the most frequent habitat association (for 11 of the 20 species) was with trees and woodland, whilst 18 of the 20 species were common or widespread species at a UK scale.
The most rapidly declining species from 1990-2014 (>60% decline, most extreme decline first) were: Satyr Pug Eupithecia satyrata, Grey Mountain Carpet Entephria caesiata, Marbled Beauty Bryophila domestica, Smoky Wave Scopula ternata, Red Carpet Xanthorhoe decoloraria, Grass Rivulet Perizoma albulata, Mottled Umber Erannis defoliaria, Juniper Pug Eupithecia pusillata, Cabbage Moth Mamestra brassicae, Currant Pug Eupithecia assimilata and Scarce Silver Y Syngrapha interrogationis. Of the top 20 species in most rapid decline, 13 were generalists, whilst the remainder were either associated with woodland and moorland habitats in equal proportions.
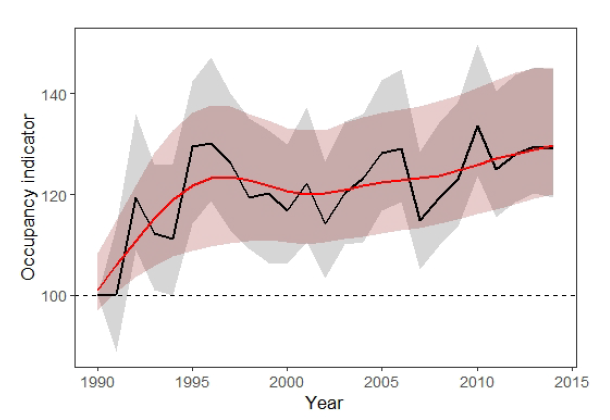
The composite indicator (based on 230 species) of moth occupancy (Figure. 2), shows an increase over both periods of assessment (by 16% 1990-2014; 5% 2005-2014). It is possible that the positive trend in the occupancy indicator is being driven in part by the range expansion of southerly distributed common and widespread species benefitting from climate warming (Morecroft et al., 2009; Conrad et al., 2006; Fox et al., 2014). There is evidence for northern range expansion in some moth species (Mason et al., 2015), which is consistent with the general pattern of poleward range expansions documented for other taxa in Britain and globally (Hickling et al., 2006; Chen et al., 2011). However, a more detailed traits analysis is needed to confirm this, specifically for Scotland.
Comparisons between occupancy and abundance
Over the period 1990-2014 comparable estimates of abundance and occupancy change were available for 164 species. There was no strong correlation between individual species abundance and occupancy change, which is not surprising given that over this period the moth abundance indicator decreased whilst the occupancy indicator increased.
It is suggested that climate change may be driving this difference, but further investigation is required to determine this and the degree of representativeness of the two data types. There was a tendency for more negative abundance trends to be of a greater magnitude, in comparison with occupancy changes. Species increasing were more likely to be increasing in occupancy, whilst species declining in abundance were just as likely to be increasing in occupancy as decreasing.

Moth conservation in Scotland
Priority species
The Scottish Biodiversity List (SBL), highlights 99 moth species that are of the highest priority for biodiversity conservation in Scotland; including 14 former UK Biodiversity Action Plan (UKBAP) range restricted species, 62 UKBAP species that were put together under a single action plan for declining common moths and a further 23 additional SBL priority species. Of the 99 species, there are 13 where conservation action on the ground is deemed necessary, including ten former UKBAP species. These are: Cousin German Protolampra sobrina, Dark Bordered Beauty Epione vespertaria, Forester Adscita statices, Goat Moth Cossus cossus, Lunar Yellow Underwing Noctua orbona, Netted Mountain Moth Macaria carbonaria, New Forest Burnet Zygaena viciae, Northern Dart Xestia alpicola, Slender Scotch Burnet Zygaena loti and Small Dark Yellow Underwing Coranarta cordigera plus the micro-moths Currant-shoot Borer Lampronia capitella, Pied Clothes Moth Nemapogon picarella and Scarce Long-horn Nemophora degeerella. The remaining 86 species are described under a ‘watching brief’ and require further survey, monitoring and/or research efforts to establish status, threats and requirements.
BCS has recently reviewed its species conservation priorities via the production of its 2025 Scottish Conservation Strategy. This identifies 96 moth species (41 macros and 55 micros) spread across three priorities. Within the list, Butterfly Conservation Scotland, has identified 13 species requiring conservation action, as part of its 2025 Conservation Strategy, including eight species not on the SBL list. The eight non-SBL species are: Barred Tooth-striped Trichopteryx polycommata, Beautiful Snout Hypena crassalis (recently discovered in Scotland), Diana's Choreutis Moth Choreutis diana, Kentish Glory Endromis versicolora, Lost Ermel Kessleria fasciapennella, Pine-tree Lappet Dendrolimus pini, Transparent Burnet Zygaena purpuralis and Welsh Clearwing Synanthedon scoliaeformis.
It should be noted that the SBL was compiled 13 years ago and is likely to be revised, whilst the Butterfly Conservation Scotland list is under development and may be subject to further changes.
A summary of the 21 species identified as requiring ‘conservation action’ (targeted land management) in Scotland (SBL and BC lists combined) is given in Table 2. Record data was not available for six species, these being New Forest burnet and five micro-moth species. For 11 of the 15 species with available record data at 1-km resolution (approximately 74% of the total), the number of records has increased in recent years; reflecting efforts by BC through the NMRS to increase recording effort. In spite of this, a robust abundance or occupancy trend is only calculable for one (New Forest Burnet) of the 21 species requiring conservation action, due to data deficiencies. There is an urgent need to address this knowledge gap, so that the effectiveness of any conservation interventions can be evaluated and reported. Of the other 20 species requiring conservation action, trends have been assessed for the 16 macro moth species by expert opinion (pers comm. Tom Prescott, BC Scotland), though there is considerable uncertainty in the assessment. One species is thought to be increasing, four species may be declining, with the remaining trends either classed as uncertain or showing no overall change.
Table 2. Species in Scotland where conservation action (land management) is planned or underway either by NatureScot and/or BC, with number of records (1km2 resolution) per decade)
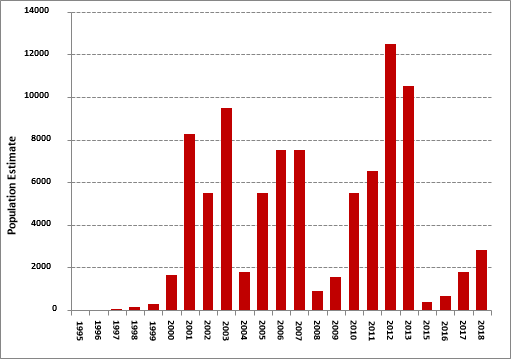
The New Forest Burnet Zygaena viciae adult population is monitored annually and vegetation approximately every three years at the sole UK site by NatureScot with assistance from BCS volunteers. Comparable measures of population size (per Mark Young and David Barbour) have been obtained from a combination of transect counts and mark-release recapture experiments. These data show large individual abundance increases in some years relative to the baseline in the mid-1990s, when the moth population was close to extinction. However, given large annual variability, including very low estimates in certain years, particularly 2008, 2015 & 2016, the linear change over time is not significant and the trend is classed as stable.
There are six former UK BAP Priorities that are not High Conservation Priorities on the SBL or BC list, chiefly due to increased recording effort; finding these species to be more widespread than previously known; either in Scotland or across the UK as a whole. These include: Argent & Sable Rheumaptera hastata, Narrow-bordered Bee Hawk-moth Hemaris tityus, Square-spotted Clay Xestia stigmatica and Sword-grass Xylena exsoleta. The actions for White-spotted Sable Anania funebris (a micro) are research and further surveys, whilst the Scarce Crimson and Gold Pyrausta sanguinalis (a micro) is thought to be extinct in Scotland, with Northern Ireland being the only part of the UK that holds this rare moth.
Conservation action on the ground
To enhance the population threatened moth species it is essential to engage with landowners and local volunteers. In most cases effective habitat management advice can only be given if the life-cycle and habitat requirements of a species are fully understood. However, for many species there are knowledge gaps, particularly in the requirements of the caterpillar stage. The series of short species summaries below briefly outlines some of the work that has been undertaken on Priority Species in recent years.
New Forest Burnet Zygaena viciae
Despite its name this day-flying moth has been extinct in England since 1927, but survives at a single site on a very remote area on the west coast of Scotland. Conservation efforts focused on excluding sheep from the site by fencing have been extremely successful, resulting in the population increasing from a low of 20 in the 1990s to a peak of around 12,500 in 2012. However, in the winter season of 2014/2015 the fence surrounding the site was breached by land-slips and falling boulders that enabled sheep access to the colony. In the summer of 2015 emergency fence repairs were undertaken by volunteers from BC Scotland’s Highland Branch and the sheep again excluded, only for further damage to the fence and ingress of sheep the following winter. The original fence was in very poor state with many loose and broken posts, snapped and brittle wires and it was deemed no longer viable to continue with further maintenance and repairs. In October 2017, a new perimeter fence was installed by a local contractor that should exclude sheep for the next twenty-five years. Given the site’s remoteness and steep terrain all materials had to be transported to site by helicopter. The new fence was erected on the inside of the existing one in the hope that it will provide some protection by catching debris from any future landslips.
Moth numbers increased in 2017 after two very poor seasons due to the ingress of sheep and inclement weather but very encouragingly monitoring in 2018 resulted in the highest count since 2013. Volunteers also assisted with a successful caterpillar survey, the first time that this has been undertaken and ongoing vegetation monitoring to help determine and understand the moth’s preferred habitat. These data are currently being analysed.
Kentish Glory Endromis versicolora
Despite its name the beautiful Kentish Glory is only found in the Cairngorms and Moray having become extinct from several English counties due to declines of the moth’s favoured ephemeral woodland habitat; short birch trees. The moth is also vulnerable due to its low powers of dispersal, few records away from two or three core sites, habitat fragmentation and resulting isolation of populations. Its steady decline leading to its extinction across several counties in England highlights its vulnerability.
Kentish Glory has become less common at one of its former strongholds just north of Aviemore, leading to concerns about its continued presence. BCS has been working closely with the landowner and are due to restructure the woodland and restrict grazing using temporary exclosures starting in the 2018/19 winter. The colonisation of the moth into newly created habitat will be monitored to help determine its success and powers of dispersal. Lessons learnt at this site will be valuable and provide confidence that similar management will succeed elsewhere. It will also enhance the site’s population allowing it to spread to adjoining areas.
The development of a pheromone lure, through collaboration with Canterbury Christchurch University, has been trialled by volunteers since 2016. This has resulted in the discovery of several new colonies and confirmation of its continued existence at other sites where it had not been seen for many years. A volunteer project also looked at the moth’s dispersal by catching, marking and recapturing individuals, using the lures. Further work is planned to enhance the pheromone as well as undertake further studies.

Dark Bordered Beauty Epione vespertaria
This very rare moth is associated with young aspen suckers and occurs at three locations in Scotland (and just one in England). Its populations are primarily monitored by volunteers. The fortunes of this moth are overseen by the Dark Bordered Beauty Steering Group, which is comprised of individuals and organisations including NatureScot, BCS, RSPB and Coille Alba. Habitat management including: coppicing taller aspen and scrub and the erection of temporary and/or permanent fencing to exclude livestock and deer been undertaken at most extant sites. The moth population has responded well at some sites but not at others, raising concerns over the lack of detailed knowledge of the moth’s ecological requirements, particularly at the caterpillar stage. To this end NatureScot have recently appointed a graduate placement to look into the ecology of this species, and assist with its annual monitoring.
Slender Scotch Burnet Zygaena loti
This day-flying moth only has four populations in the UK, all of which are on Mull and the adjacent island of Ulva. Concerns have grown recently over a lack of recording and annual monitoring of the moth, particularly away from one of its strongholds at Ardmeanach where it is managed and monitored by the NTS. In 2018 BCS held a workshop, part funded by NatureScot, at Glengorm Estate to encourage local volunteers to survey and monitor this species and its cousin Transparent Burnet on the island. In addition a Glasgow University student undertook a study of the moth, looking at population density in relation to habitat attributes and also the moth’s dispersal.
For several years BCS has held volunteer work parties at Kilninian, on the west coast of the island, and one of the moth’s former strongholds to clear alien and invasive cotoneaster that has slowly smothered the moth’s preferred habitat (species-rich undercliffs). It is encouraging that suitable habitat is returning although the moth has not been recorded for a few years.
Forester Adscita statices
This metallic green day-flying moth has a very restricted distribution in Scotland, only occurring in parts of Argyll and adjacent islands, and in Dumfries and Galloway. The adults are found in damp meadows and flower-rich dry grassland, although the moth is not known from several sites that appear to have suitable habitat. In an attempt to find out more, BCS held a training day part funded by NatureScot in Kilmartin where local volunteers were encouraged to search for caterpillars and their feeding signs. This resulted in the discovery of feeding signs at two sites, the first time they have been found in the wild in Scotland. Hopefully this will help to unlock crucial information about the moth’s habitat requirements in Scotland.

References
Agassiz, D.J.L. 2004. Cypress trees and their moths. British Wildlife, 15, 265-268
Buckland, S.T., Magurran, A.E., Green, R.E. & Fewster, R.M. 2005. Monitoring change in biodiversity through composite indices. Phil. Trans. R. Soc. B, 260, 243-254.
Burns, F., Eaton, M.A., Barlow, K.E., Beckmann, B.C. et al. 2016. Agricultural management and climatic change are the major drivers of biodiversity change in the UK. PLoS ONE, 11(3), e0151595.
Chen, I., Hill, J.K., Ohlemüller, R., Roy, D.B. & Thomas, C.D. 2011. Rapid range shifts of species associated with high levels of climate warming. Science, 333, 1024-1026.
Conrad, K.F., Warren, M., Fox, R., Parsons, M. & Woiwod, I.P. 2006. Rapid declines of common, widespread British moths provide evidence of an insect biodiversity crisis. Biological Conservation, 132, 279-291.
Dennis, E.B., Morgan, B.J.T., Freeman, S.N., Ridout, M.S., Brereton, T.M., Fox, R., Powney, G.D. & Roy, D.B. 2017. Efficient occupancy model-fitting for extensive citizen-science data. PLoS ONE, 12(3), e0174433.
Dennis, E.B., Brereton, T.M., Morgan, B.J.T., Fox, R., Shortall, C.R., Prescott, T. & Foster, S. 2019. Trends and indicators for quantifying moth abundance and occupancy in Scotland. Journal of Insect Conservation, in press.
Fox, R. 2013. The decline of moths in Great Britain: a review of possible causes. Insect Conservation and Diversity, 6, 5-19.
Fox, R., Oliver, T.H., Harrower, C., Parsons, M.S., Thomas, C.D. & Roy, D.B. 2014. Long-term changes to the frequency of occurrence of British moths are consistent with opposing and synergistic effects of climate and land-use changes. Journal of Applied Ecology, 51, 949-957.
Fox, R., Parsons, M.S., Chapman, J.W., Woiwod, I.P., Warren, M.S. & Brooks, D.R. 2013. The State of Britain’s Larger Moths 2013. Butterfly Conservation and Rothamsted Research, Wareham, Dorset.
Hahn, M. & Brühl, C.A. 2016. The secret pollinators: an overview of moth pollination with a focus on Europe and North America. Anthropod-Plant Interactions, 10, 21-28.
Hassall, C. & Thompson, D.J. 2010. Accounting for recorder effort in the detection of range shifts from historical data. Methods in Ecology and Evolution, 1, 343-350.
Heath, J. & Emmet, A.M. 1983. The Moths and Butterflies of Great Britain and Ireland. Harley Books, Colchester.
Hickling, R., Roy, D.B., Hill, J.K., Fox, R. & Thomas, C.D. 2006. The distributions of a wide range of taxonomic groups are expanding polewards. Global Change Biology, 12, 450-455.
Hill, L., Randle, Z., Fox, R. & Parsons, M. 2010. Provisional Atlas of the UK’s Larger Moths. Butterfly Conservation, Wareham.
Isaac, N.J.B., van Strien, A.J., August, T.A., de Zeeuw, M.P. & Roy, D.B. 2014. Statistics for citizen science: extracting signals of change from noisy ecological data. Methods in Ecology and Evolution, 5, 1052-1060.
Isaac, N.J.B. & Pocock, M.J.O. 2015. Bias and information in biological records. Biological Journal of the Linnean Society, 115, 522-531.
Leverton, R. 2005. Chamomile Shark Cucullia chamomillae in north-east Scotland. Atropos, 26, 53-54.
Leinonen R., Söderman G., Itämies J., Rytkönen S. & Rutanen I. 1998. Intercalibration of different light-traps and bulbs used in moth monitoring in northern Europe. Entomologica Fennica, 9, 37-51.
Macgregor, C.J., Pocock, M.J.O., Fox, R. & Evans, D.M. 2015. Pollination by nocturnal Lepidoptera, and the effects of light pollution: a review. Ecological Entomology, 40, 187-198.
Mason, S.C., Palmer, G., Fox, R., Fillings, S., Hill, J.K., Thomas, C.D. & Oliver, T.H. 2015. Geographical range margins of many taxonomic groups continue to shift polewards. Biological Journal of the Linnean Society, 115, 586-597.
Morecroft, M.D., Bealey, C.E., Beaumont, D.A., Benham, S., Brooks, D.R., Burt, T.P., Critchley, C.N.R., Dick, J., Littlewood, N.A., Monteith, D.T., Scott, W.A., Smith, R.I., Walmsley, C. & Watson, H. 2009. The UK Environmental Change Network: emerging trends in the composition of plant and animal communities and the physical environment. Biological Conservation, 142, 2814-2832. Scottish Natural Heritage. 2011. Trend Note—Scottish Moths. Accessed Aug 2018
Storkey, J., Macdonald, A.J., Bell, J.R., Clark, I.M., Gregory, A.S., Hawkins, N.J., Hirsch, P.R., Todman, L.C. & Whitmore, A.P. 2016. The unique contribution of Rothamsted to ecological research at large temporal scales. In: A J. Dumbrell, R L. Kordas & G. Woodward, eds. Advances in ecological research (Vol. 55, pp. 3–42), Chapter 1. Cambridge: MA: Academic Press.
Taylor, L.R. & French, R.A. 1974. Effects of light-trap design and illumination on samples of moths in an English woodland. Bulletin of Entomology Research 63, 583-594.
Tingley, M.W. & Beissinger, S.R. 2009. Detecting range shifts from historical species occurrences: new perspectives on old data. Trends in Ecology & Evolution, 24, 625-633.
Williams, C.B. 1948. The Rothamsted light-trap. Proc R Entomol Soc Lond A 23,80-85.
Williams, C.B. 1952. Some notes on killing insects for collections and for scientific research. Entomologist 85, 271-279.
Woiwod, I.P. & Harrington, R. 1994. Flying in the face of change: The Rothamsted Insect Survey. In: Leigh RA & Johnston AE (eds) Long-term experiments in agricultural and ecological sciences, CAB International, London, pp 321-337.
Disclaimer: Scottish Natural Heritage (SNH) has changed its name to NatureScot as of the 24th August 2020.
At the time of publishing, this document may still refer to Scottish Natural Heritage (SNH) and include the original branding. It may also contain broken links to the old domain.
If you have any issues accessing this document please contact us via our feedback form.






